The DOH’s approval of the Cepheid test kit brings us closer to nationwide mass testing, but there’s still so much to do.
The Food and Drug Administration (FDA), under the Department of Health, has approved three new testing kits for commercial use in the Philippines in a statement released on Thursday, March 26. Coming to a total of 15 FDA recognized COVID-19 test kits in the country, the announcement’s significant point is the approval of the Cepheid testing kit.
.
The situation now
Current testing methods available in the country’s system can achieve diagnostic results in around 48 hours, as well as being only done in select hospitals in the country. At the start of the outbreak, only the Research Institute for Tropical Medicine (RITM) in Muntinlupa was the only testing site for diagnosis, meaning all samples were sent to just this one site. But since the rapid spread of the virus, other hospitals and clinics have been approved as testing sites including San Lazaro Hospital (Manila), Baguio General Hospital and Medical Center (Benguet), Vicente Sotto Memorial Medical Center (Cebu), and Southern Philippines Medical Center (Davao)–but still a very limited number of test sites compared to the number of PUIs.
.
Cepheid’s caveat

The Cepheid testing kit remedies both issues in time and limited test sites with its approval. With this new test kit, the lead time is greatly reduced by only needing less than an hour to yield results, currently the fastest test for diagnosing the virus. This means a faster turnover for PUIs and more accurate estimates on the virus’ rate of spreading. Another advantage of the Cepheid kit is its use of Gene Xpert, the same machine used for TB testing, a tool that is already in many medical facilities nationwide. This means that no new equipment will have to be incurred for hospitals and clinics to implement this new testing kit and diagnosis won’t be constrained to a few test sites. This makes mass testing for the virus a greater possibility in the country.
.
Also read: How To Aid Filipino Healthcare Workers And Front Liners During The COVID-19 Pandemic
.
With all the benefits that come with the approval of the Cepheid kit, it also comes with a new set of hurdles. The approval of Cepheid doesn’t mean the Philippines will have immediate access to the kits. Produced in the US, the limited number of kits produced poses a supply problem as the US is also buying the kit’s cartridges for their own testing. Price is also an issue in acquiring a bulk of Cepheid’s COVID-19 cartridges, priced at an estimate $20 per cartridge compared to the estimate price of TB cartridges at $10. These prices don’t even account for shipping and other costs that will be added on when the kit reaches the Philippines. Without additional monetary support or intervention, it may reach months until the government will have access to enough Cepheid cartridges to properly implement nationwide mass testing.
.
Local alternatives
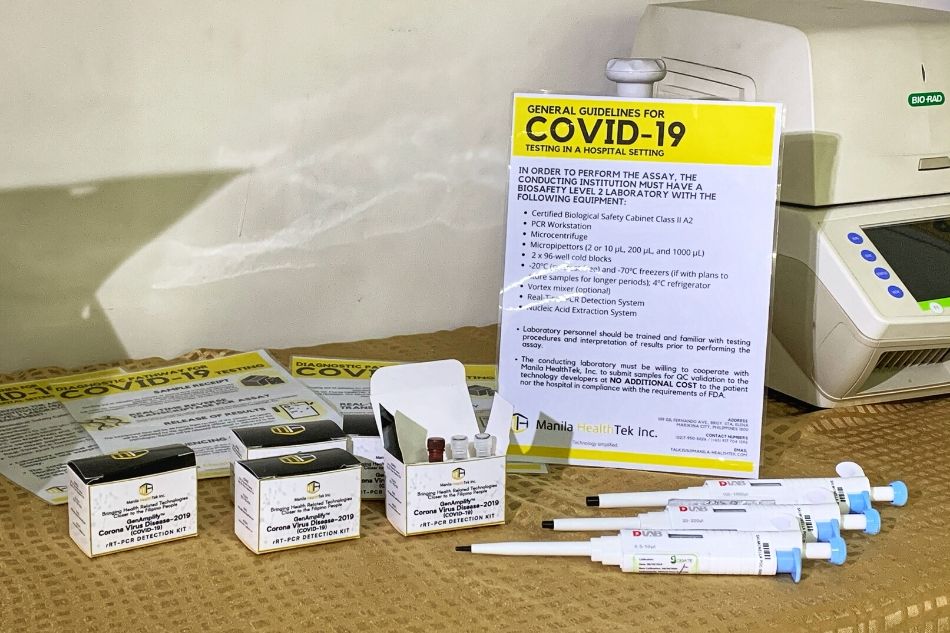
Alternatively, there are other less costly testing methods available to us. The locally made test kit, GenAmplify, by Dr. Raul Destura and his team at the University of the Philippines Manila-National Institutes of Health (UP-NIH) is our strongest bet in achieving mass testing locally. Though their test kit is still under evaluation by the FDA, which will take around two weeks, Dr. Destura’s team still needs support for research and testing to fully equip the kit for FDA approval and to produce enough test kits to meet the demand from hospitals, at the current rate of production they can only produce around 200 kits a week. This local alternative must be considered as a viable option because over-relying on foreign testing is too expensive and risky. Just recently, the DOH received and allocated 100,000 test kits donated from China. These procured test kits may appear to be faulty with reports from Spain stating that only 30% of the test kits they bought from China properly identified the virus. The same with the Czech Republic with a report that 80% of their China sourced test kits produced erroneous results.
.
The search for a cure
All of these developments are only addressed in diagnosing the virus in patients. The research into finding a cure or even a vaccine for COVID-19 is still in progress, but the World Health Organisation (WHO) has already started a global initiative into finding a treatment for infected patients. Called the SOLIDARITY trial, the WHO has identified four drugs to test on infected patients to slow or kill the novel coronavirus. Namely, these drugs are: Remdesivir, a compound that shuts down viral replication; Chloroquine and hydroxychloroquine, a drug duo that inhibits a virus from entering and infecting healthy cells; Ritonavir/lopinavir, another combination drug that inhibits the protease of viruses; and Ritonavir/lopinavir and interferon-beta, is a trial that uses the previous drug with added interferon-beta, which is used to regulate inflammation in patients. The nature of the SOLIDARITY trial can change over time as more information is collected from the clinical trials of these drugs. For now, proper detection is what is needed to control the outbreak locally.
.
How to help now
What all of this boils down to support, we need to give what we can to flatten the curve of the virus within our country. Donating to Dr. Destura’s initiatives can greatly hasten the approval and shipment of the GenAmplify test kits across the country. Purchasing the Cepheid kits for donation can also help in quickly detecting the virus in PUIs. Of course, giving aid to front liners and healthcare workers will also greatly strengthen our plight in controlling the virus.
To support GenAmplify, you can deposit your cash donation to:
The U.P. Medical Foundation, Inc. Bank:
BDO, Bocobo-Pedro Gil Branch
Account No.: 00-353-0094-018
Swift Code: BNORPHMM
For more information on Cepheid test kits, check their website for more information.
For donations to Filipino front liners and healthcare workers, read more here.
Featured image from Cepheid





